Welcome to my blog on red light therapy for the gut microbiome.
Before we start, other blogs that you might be interested in, include:
- Grounding: What Is It? Why Do It? How Do It?
- Red light therapy for thyroid function.
- Red and Near-Infra Red Light Therapy
- Rhodiola Health Benefits.
- Ashwagandha: One Of My Favourite Supplements
How Does Red Light Therapy For The Gut Microbiome Work?
The mechanisms of action of red light therapy have been widely investigated in recent years, and additional mechanistic information is still being discovered.
Nevertheless, it is generally accepted that the single most important chromophore in the red and near-infrared regions of the spectrum is cytochrome c oxidase (CCO).
A chromophore is the part of a molecule responsible for its color.
Cytochrome c oxidase (CCO), is unit IV of the mitochondrial respiratory chain – our mitochondria are a sub-component of all of our cells and the site of energy production. Mitochondria used to be bacteria (1.5 billion years ag0) and we now understand they sense, integrate, and orchestrate human health.
Watch this interview with Ray Griffiths to learn more about our mitochondria:
When CCO absorbs light, the enzyme activity is increased leading to:
- Increased electron transport.
- More oxygen consumption.
- Higher mitochondrial membrane potential.
- Increased ATP production.
How Does Red Light Therapy Effect The Gut?
We have different levels of evidence, most coming from animal research:
- Circadian rhythm changes.
- Vitamin D.
- Immune modulation (anti-inflammatory)
The effect of the circadian rhythm on the microbiome has been demonstrated and the bacteria responsible for decreased gut integrity and increased lipopolysaccharide transport are upregulated in mice after disruption of the sleep/wake cycle.
In addition to circadian rhythm, light also has an indirect effect on the microbiome through vitamin D, produced by the action of sunlight on keratinocytes. Vitamin D is known to boost immune function by the induction of antimicrobial peptide genes and the regulation of tight junction proteins in the epithelial layer of the intestine and to maintain microbiome homeostasis and protect against colitis in mice, possibly by controlling inflammation.
The alteration of the microbiome, that was observed in mouse experiments, may be due to a secondary effect of PBM, affecting the mouse inflammatory response, and in turn affecting the gut microbiome (1).
This is entirely feasible, given the intimate relationship between the microbiome (healthy and dysbiosis) and the inflammatory response. It is hypothesised that this effect may be due to the well-known anti-inflammatory and redox signalling effect of PBM.
PBM can reduce pro-inflammatory cytokines, such as IL-6, TNF-α, IFN-γ, and change the activity of macrophages and neutrophils. (1)
The Gut Brain-Axis and Red Light Therapy
Red light therapy for the gut microbiome could remediate mitochondrial dysfunction in gut neurons, reinstating the complex bidirectional communication system between the enteric nervous system and the central nervous system (the gut/brain axis). (1)
This may have particular significance for neurodegenerative conditions, such as Alzheimer’s and Parkinson’s diseases, both of which involve early pathological abnormalities in the gut/brain axis.
Another theory the authors laid out was that the local effect of red light therapy on inflammatory pathways may have systemic consequences:
It is possible that circulating immune cells (mast cells, macrophages, etc.), stimulated by red light therapy, could transduce protective signals from distal tissues to sites of injury such as the brain, heart, or gut. (1)
The authors of this paper have shown in a previous study that red light therapy, delivered as low-level laser, to the abdomen of healthy mice can produce a significant change in the gut microbiome.
Red light therapy significantly altered the microbial diversity of the microbiome, an effect most pronounced in mice treated three times per week with NIR light (808 nm). (1)
Red Light Therapy Increases Keystone Species Of The Gut Microbiome
Recent preliminary work from the authors laboratory (unpublished research) has also indicated that red light therapy for the gut microbiome in humans improves diversity as well as increasing keystone species such as Akkermansia muciniphila, Bifidobacterium sp., and Faecalibacterium sp.. All recognised as being correlated with a healthy microbiome, and decreases in the Firmicutes:Bacteroides ratio, proposed as an indicator of gut health.
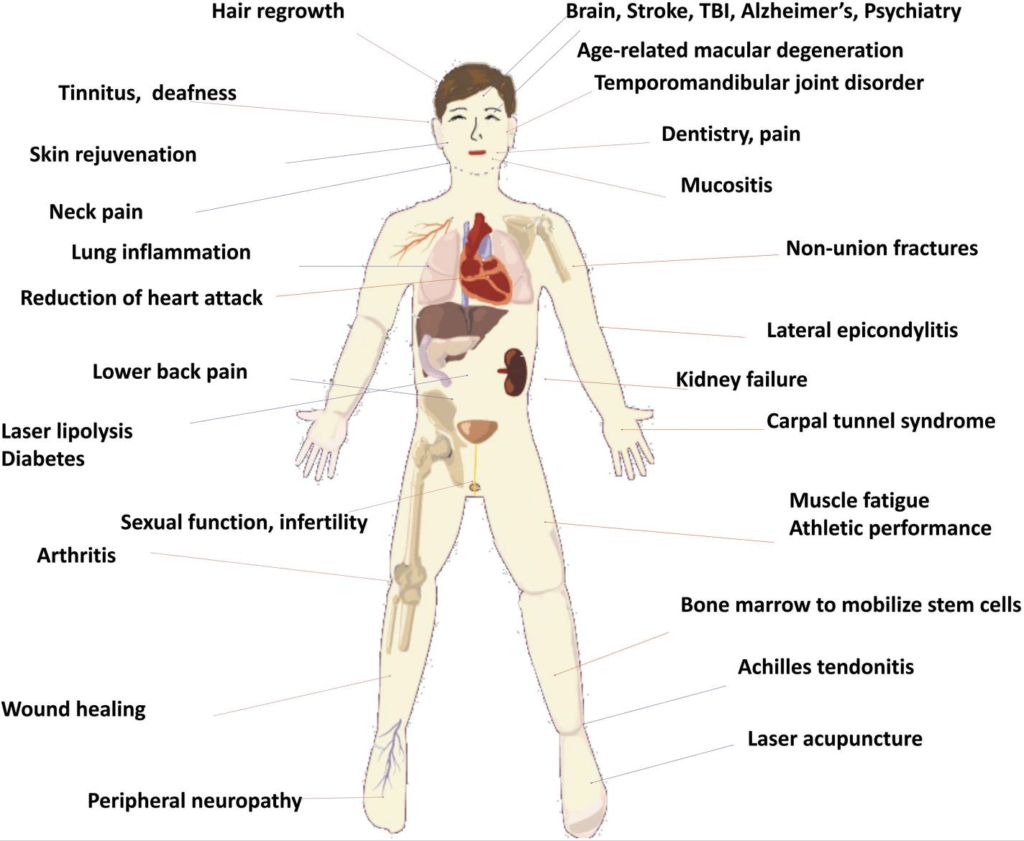
Schematic illustration of the wide variety of human diseases and disorders that have been treated by PBM. (1)
Conclusions
Red light therapy for the gut microbiome may serve as a way to beneficially change the microbiome for a number of different inflammatory and neurological diseases (such as cardiovascular and Parkinson’s diseases).
The obvious approaches to try to improve the microbiome in humans such as diet, probiotics, and fecal transplants have had some success, but these treatments do not amount to a complete solution for the entire problem. Fecal transplants are currently being used for Clostridium difficile infection, irritable bowel disease, ulcerative colitis, and are also being considered for some non-intestinal metabolic diseases.
A healthy microbiome would balance SCFA production, serotonin/kynurenine pathways, trimethylamine metabolism, and dopamine and neurotransmitter production, which, in turn may affect the outcome of some of the most difficult-to-treat diseases, including Parkinson’s disease, multiple sclerosis, amyotrophic lateral sclerosis, attention-deficit/hyperactivity disorder, and autism spectrum disorder.
References
- “Photobiomics”: Can Light, Including Photobiomodulation, Alter the Microbiome? (click here)
Alex is a certified Functional Medicine Practitioner (IFMCP) and has a MSc in Personalised Nutrition. He is also a breathwork facilitator with a background in personal training and massage therapy. He also runs The Resiliency Program - a 24 week program aimed at building physical, mental, emotional, and spiritual resilience.