Welcome to my blog post ‘The Gut-Thyroid Axis In Thyroid Disease’ where I discuss some of the research exploring the connection between our gut microbiota and thyroid function.
You may also be interested in the section of my blog dedicated to gut health, click here, in particular:
- The Gut Microbiome In Multiple Sclerosis: What Bacteria Are Involved
- Psychobiotics: Probiotics For Mood And Mental Health
What Is The Gut-Thyroid Axis?
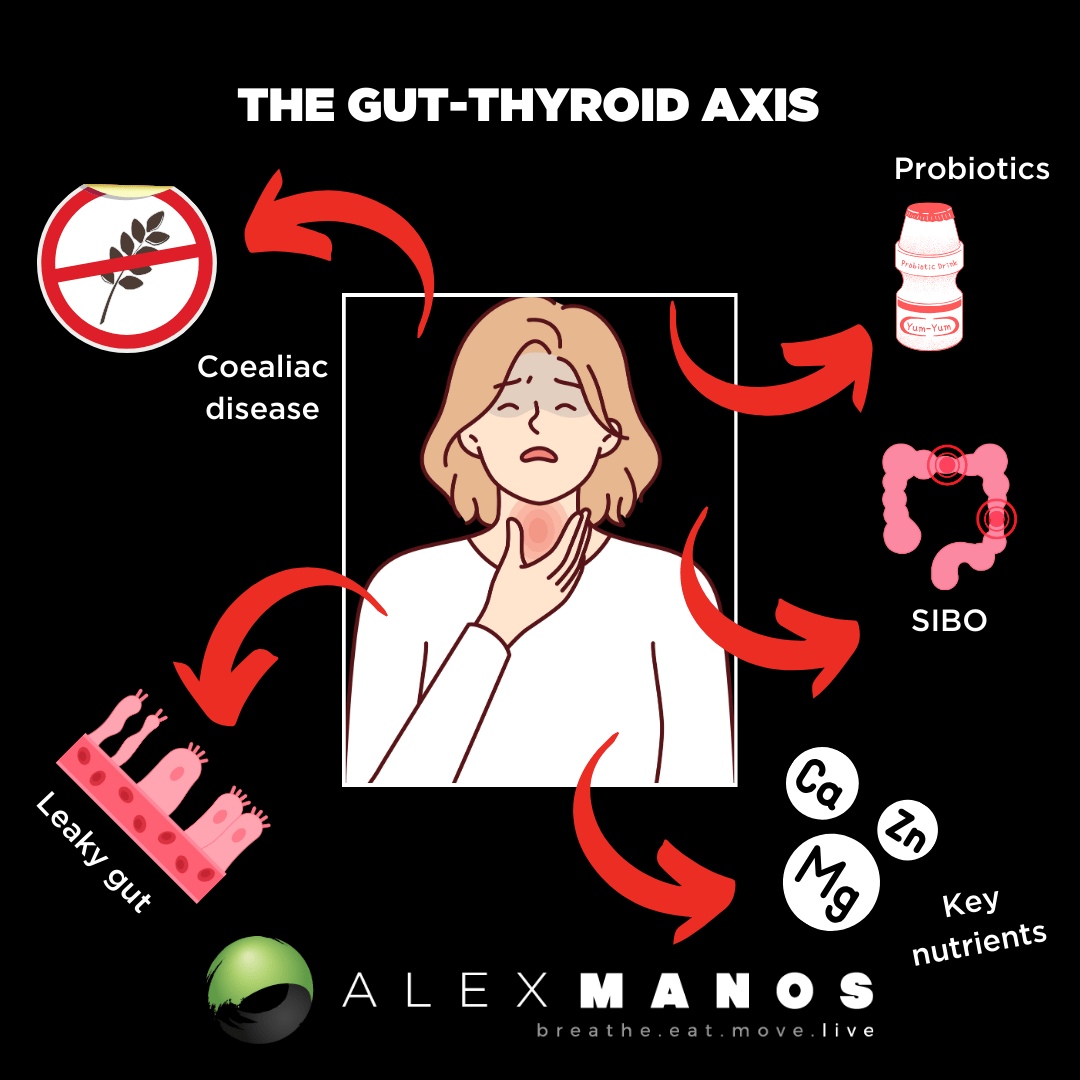
A healthy gut microbiome not only has beneficial effects on the activity of the immune system, but also on thyroid function. Thyroid and intestinal diseases prevalently coexist—Hashimoto’s thyroiditis (HT) and Graves’ disease (GD) are the most common autoimmune thyroid diseases (AITD) and often co-occur with Celiac Disease (CD) and Non-celiac wheat sensitivity (NCWS) (1).
How Does The Gut Cause Thyroid Disease?
This can be explained by the damaged intestinal barrier and the following increase of intestinal permeability, allowing antigens to pass more easily and activate the immune system or cross-react with extra-intestinal tissues, respectively. An imbalance in the microbiome has not only been found in autoimmune thyroid diseases, but has also been reported in thyroid carcinoma, in which an increased number of carcinogenic and inflammatory bacterial strains were observed.
Additionally, the composition of the gut microbiota has an influence on the availability of essential micronutrients for the thyroid gland. Iodine, iron, and copper are crucial for thyroid hormone synthesis, selenium and zinc are needed for converting T4 to T3, and vitamin D assists in regulating the immune response. Those micronutrients are often found to be deficient in AITDs, resulting in malfunctioning of the thyroid (1).
Iodothyronine-deiodinases play an important role in the conversion of thyroxine (T4) to its active form T3 or reverse T3, its inactive form. Deiodinase activity has also been found in the intestinal wall and could contribute to total T3 body levels.
Another influencing factor of the microbiome is its effect on neurotransmitters such as dopamine, which can inhibit thyroid stimulating hormone (TSH).
The microbiome influence the uptake of minerals relevant to the thyroid including iodine, selenium, zinc, and iron.
All of them play a role in supporting thyroid function and there is a clear link between thyroid dysfunction and altered levels of these minerals.
Beyond that, there seems to be a negative correlation between Lactobacillaceae and Bifidobacterium spp. with dietary iron and a positive correlation with selenium and zinc. As these bacteria are diminished in Hashimoto thyroiditis and Grave´s disease, it has been suggested that gut composition and mineral regulation may have an impact on these diseases (1).
Do Probiotics Help Thyroid Disease?
Supplementation of probiotics showed beneficial effects on thyroid hormones and thyroid function in general.
Watch my interview with Dr. Joe Mather from The Ruscio Institute:
Interestingly, microbes like E. coli function as a reservoir for T3 by binding it to bacterial thyroid-binding hormone and are able to prevent thyroid hormone fluctuating and thus, possibly reduce the need for T4 supplementation. Spaggiari et al. investigated the influence of Lactobacilli and Bifidobacteriaceae probiotics on levothyroxine. They found a significantly lower adjustment requirement of T4 in the study compared to the control group, reasoning that microbiota modification increases levothyroxine availability and stabilizes thyroid function. They concluded that probiotics have a beneficial role in lowering serum hormone fluctuations, also considering that iodothyronines deconjugation is regulated by bacterial enzymes sulfatases and ß-glucuronidases, which could be more available due to probiotics.
Probiotics seem to be able to accumulate trace elements such as selenium, zinc, and copper and incorporate them into organic compounds. Considering that selenium, zinc, and probiotics operate via different pathways and all of them are favorable for the thyroid, there could be a synergistic effect for health when incorporating all of them, especially in deficient conditions.
Probiotics could constitute an adjuvant therapy for thyroid diseases.
Summary of ‘The Gut-Thyroid Axis In Thyroid Disease’
- There is accumulating data that a strong thyroid–gut axis exists.
- It appears to display a not well known but important correlation regarding the effect of the gut bacteria on the immune system and thyroid function.
- There is higher prevalence of the coexistence of thyroid and gut related diseases, just as Hashimoto’s Thyroiditis/Graves’ Disease and Celiac Disease/Non-celiac wheat sensitivity.
- Dysbiosis is a common finding in thyroid disorders.
- On the one hand, it alters the immune response by promoting inflammation and reducing immune tolerance, damaging the intestinal membrane and causing an increase in intestinal permeability, which again leads not only to a high exposure of antigens, but also local inflammation.
- On the other hand, it can directly impact thyroid hormone levels through its own deiodinase activity and the inhibition of TSH.
- Gut microbiota also influences the absorption of minerals that are important to the thyroid, including iodine, selenium, zinc, and iron. All of them are essential for thyroid function and there is a clear link between thyroid dysfunction and altered levels of these minerals.
- Probiotics have shown beneficial effects in thyroid diseases and are able to have a positive effect on trace elements such as selenium, zinc, and copper. Additionally, microbes function as a reservoir for T3 and are able to prevent thyroid hormone fluctuating and thus may be able to reduce the need for T4 supplementation.
- Probiotics could constitute an adjuvant therapy for thyroid diseases. However, most studies on probiotics rely on animal models, therefore well-designed human studies are needed to further elucidate the importance of the thyroid–gut axis and the possibilities of intervention.
References of ‘The Gut-Thyroid Axis In Thyroid Disease’:
- Thyroid-Gut-Axis: How Does the Microbiota Influence Thyroid Function: click here.
Alex is a certified Functional Medicine Practitioner (IFMCP) and has a MSc in Personalised Nutrition. He is also a breathwork facilitator with a background in personal training and massage therapy. He also runs The Resiliency Program - a 24 week program aimed at building physical, mental, emotional, and spiritual resilience.